Part 1 - Gas.
Gases boil at different temperatures. When a gas goes above it's 'boiling point' it will change state from a liquid to a vapour. Our stoves require a minimum pressure difference between the outside ambient air pressure and the pressure inside the canister so we get a good flow of gas to the burner. As a rough guide you want the gas temperature to be about 10C above the boiling point of that gas.
We typically use three different gases in New Zealand for outdoors stoves. They are n-Butane , Isobutane and Propane. Pure gases. Isobutane is the same atoms of n-butane rearranged which changes it's properties. Note: These are all Liquified Petroleum Gases. What we buy for BBQ's in New Zealand is typically a mix of Propane:Butane with a ratio of 60:40.
The boiling points of these gases at sea level are:
n-Butane: -1C
Isobutane:-11.7C
Propane:-42C
Pressure is a function of the volume of the container and the temperature of the gas. So, let's imagine we could buy three identical volume canisters from the store each containing one of these three gases. It's a real cold morning, -1C. We sit the gas canisters outside for an hour and they're now at -1C also. If the volume is the same and the temperature is the same they'll all be the same pressure right? No. The temperature of the gases is all the same at -1C but the temperature of the gases above their individual boiling points is different. -1C is a damn hot day to Propane. You try get your stove to light with that n-Butane canister and their is no pressure inside to push any gas out.
So why can't we buy Propane in those handy disposable canisters we all use for hunting if it's so great for cold temperatures? The canisters are manufactured to a European standard called EN417. EN417 outlines specifications for disposable gas canisters 50ml-1000ml. It states at 50C the maximum pressure must be no greater than 13.2bar. Propane reaches that around 40C at sea level and it's a steepening curve as can be seen in the below picture.
You can buy 100% propane however but it will be in a heavier duty container and you'll notice it will have stronger thicker walls, welded joins and a safety pressure relief valve.It will also have a different connection on it, probably a CGA600 connector.
Our little EN417 canisters are very thin and typically crimped at the top and bottom and we have what is called a Lindal valve for connection to our stoves. Below is an image of a canister I've opened up so we can see how it's made.
Yellow circle: Lindal valve
Green circles: Seals
Red circles: Crimps
Notice how the base of the canister is inverted? EN417 requires that base to overt before the canister ruptures. They do overt but in reality they often overt and milliseconds later the canister explodes so you wouldn't rely on it to overt and warn you in time it's going to explode soon.
What we are going to be buying for hunting is a mixture of Isobutane and Propane. The mix ratio will depend on the brand. MSR IsoPro gas for example is 80/20 Isobutane and Propane. It is an excellent cold weather gas with a boiling point of approximately -22C.
So, takeaways from this section : n-butane no good for colder temps - probably the cheapest gas at the store. Propane is high pressure but good for very cold temps but requires a different stove to connect to the bottle. Isobutane mixes are the best for our typical hunting trips where we are carrying our stoves and gas - different brands have different mixes.
Next I'll start showing you the different styles of stoves and some limitations of each type and some techniques to get around (to a point) these limitations.
Welcome guest, is this your first visit? Create Account now to join.
Welcome to the NZ Hunting and Shooting Forums.
Search Forums
User Tag List
+ Reply to Thread
Results 1 to 15 of 55
Thread: All about gas stoves.
-
12-04-2020, 11:14 AM #1
All about gas stoves.
-
-
12-04-2020, 11:21 AM #2Banned

- Join Date
- Mar 2015
- Location
- North Canterbury
- Posts
- 5,462
Hurry up with the next installment ! (please)
-
12-04-2020, 11:31 AM #3
Back in my younger days we exploded a few gas canisters, we sat one on a hexamethaline (sp?) cooker waiting for it to explode. The bottom overted and it fell over. Wondering what to do my mate shot it with a .243.
Word of warning, when shot they fly off in the direction the valve is pointing. The canister came flying past my mates shoulder at a rate of knots.
-
12-04-2020, 12:01 PM #4sneakywaza I got

- Join Date
- May 2013
- Location
- Fairlie
- Posts
- 3,636
-
12-04-2020, 12:09 PM #5
years back I bought a spider type gas cooker...its sstill going strong now...I see the type is now being made again...simple frame to hold simple gas head,with flexi rubber hose about 12" long to attach to screw in canister with threaded top...beauty of this system is that on a cold day or at high altitude you can place the canister close to heat and give it a bit of a boost....always thought it would be a real Mcgyver get out of jail card to sit canister ontop and run like hell before it blew up... used this for yearsand the only time it let me down was when it got soaked with rain overnight...took gas head apart and blew gauze out ,reassembled and it went fine....sometimes simple is good....I commented on this to wife the other night when watching the Duleys show......we have almost gone full circle...fire n billy...white spirits...single use gas canisters...screw on gas canisters...white spirit stoves and now back to canisters LOL. hexi cookers fit in there somewhere between fire and gas canisters and the beer can cooker rage close to end....some cheap buggas like myself never gave up on them.
-
12-04-2020, 12:45 PM #6
Part 2 - Upright canister stoves.
OK, Im going to start running through these stoves in order of worst in cold weather to best. I'm going to use MSR stoves as examples for no reason other than they have every type of stove I'm going to talk about. I am not infering they are better or worse than any other brand.
I should also add now before it's too late that I am not a chemical engineer, a mechanical engineer or a stove designer. I'm just a fella who's damn stoves wouldn't work properly when he was tahr hunting and wanted to find out why. All information I share is what I've interpreted information available on the internet to mean. If you feel anything I post is incorrect or doesn't make sense I'm OK with being wrong, we'll find the correct answer together and amend the posts.


These are both upright canister stoves. They're called that because the canister is used in an upright position only. The one with the hose we would call an upright remote canister stove.
Most of us have got or used one of these types of stoves I would say. Have you ever noticed on a cold morning your stove starts off OK but it slowly dies down and then goes out or only has a tiny flame? You shake the canister and it's got plenty of liquid sloshing around in there so what's going on?
When we use a canister in the upright position we are drawing off gas vapour from the top of the canister. As we draw that vapour off the liquid gas boils some off to replace it. That boiling or evaporating requires a heat source. Think of a pot of water on your cook top at home, it requires heat to change from a liquid to a vapour.
So where does this heat that vaporisation requires come from? The surroundings. It will actually take heat from the liquid itself. Your liquid gas is cooling down from within the longer you run the stove. We know when temperature of our gas drops our pressure drops also. That's why our stove dies off even if we have plenty of fuel. This is one of the limitations of these stoves. How do we get around that? Well you need an external source of heat for that gas. One way we all know is to start off with a warm canister. It'll still reduce in output the longer we leave the stove running but it'll probably get us a cup of coffee.
Another way if we're going to be using the stove for any greater length of time is to put a small amount of water in the pot initially and start the stove. As the stove starts to fade we turn it off, pour the warm ( not hot) water into a container and sit the canister in that. Restart the stove and now your canister has a good source of heat it'll burn much better. Always remember, it's the gas temperature relative to it's boiling point that matters. Liquid water at 5C is warm for a gas that has a boiling point of -22C. It doesn't have to feel warm to your skin necessarily (better if it does though).
The other issue with evaporation inside the canister is that the mix ratio of our gas is going to get worse towards the end of the canister. That 'enough for one night' canister you have may leave you going hungry if you take it out on a cold overnighter.
These stoves will either be using a needle type valve and a jet or a micro regulator to control the flow of gas. I'm going to come back to this after we've run through basics of each type first because it gets a bit complex and I haven't thought how to write that just yet.
-
12-04-2020, 12:51 PM #7
2nd one is similar to what I had but my hose was rubber and longer....and whole set up was lighter and less complicated LOL.
-
12-04-2020, 01:02 PM #8Member

- Join Date
- May 2018
- Location
- Porirua
- Posts
- 1,342
When you have liquid precent in the can the pressure is directly related the temperature, not the amount of fuel in the can. As @K95 says as the can empties there is less mass/heat so it ices up faster.
One more point is the boiling point is at atmospheric pressure at sea level.
Keep up the good work, looking forward to more.Remember the 7 Ps; Pryor Preparation Prevents Piss Poor Performance.
-
12-04-2020, 01:34 PM #9
Part 3 - Inverted liquid feed canister stoves.
So how do we get around those issues of evaporative cooling and our gas mix changing for the worse that we saw with the upright canister stoves? We invert the canister.
Now there is no evaporation happening inside the canister, we are drawing liquid gas off to our stove and because of that we have no internal cooling of the liquid reducing our pressure and we are drawing off the gas at the same mixture ratio for the whole canister. Win!
But( there's always a but) we need a special type of stove to do this. We need a stove that has a way of changing our liquid to a vapour before it reaches the burner. Take a look at the stove below. Notice what the green arrow is pointing to. It's a preheat loop. With this type of stove we start the stove with the canister in the upright position and run it for 30 seconds to warm up that preheat loop then we can invert the canister to liquid feed mode and the liquid will travel down the hose and hit that hot loop. When it reaches that hot loop it will evaporate and we have gas vapor for our burner.
The other advantage of this type of stove is it can be used down to much lower temperatures. When we use it in the upright canister position our valve at the canister controls the volume of fuel which is vapour. When we invert the canister and feed liquid our valve still controls the volume of fuel but the mass of our fuel has greatly increased. In short, (and I want to keep it kinda short) we can continue to get good output in liquid feed mode with very low canister pressure because of this.

Always consult a manual to see if your stove is capable of being used in liquid feed mode. Just because it has a preheat loop does not also necessarily mean it's good to use with a canister.
-
12-04-2020, 01:58 PM #10
Part 4 - Multi-fuel stoves.
All that stuff about our canister pressure being dependant on the gas temperature is a hassle isn't it? I wish we could just not worry about the temperature and make our own pressure!

Introducing the OG of cold weather stoves. The multi-fuel stove.
With this type of stove we're going to be using a fuel that will not boil off (very much) at normal temperatures we are likely to encounter. Because it's not going to boil off and it's not going to create it's own pressure inside that container. We are going to have to create the pressure we need using a manual pump located on the end of the bottle. You'll also notice it shares similarities with the last stove we talked about. See that preheater loop? Same deal expect this time we are going to pump the bottle up to pressure then open the valve and let a small amount of liquid fuel through into that bowl on the bottom of the stove. We'll close the valve again and then ignite the fuel. It's going to burn and make a tallish flame which is going to heat up our preheat loop. As the flame starts to fade we slowly open our valve again and now the liquid fuel flows down the hose and vaporises when it goes through the hot preheat loop. The vapour fuel then flows out and is ignited by the flame. We have a functioning stove now.
Multi-fuel stoves are what hard-core polar explorers use for good reason. They'll work in temperatures that will kill a human. They have limitations like any system of course but the most important of these for us hunters is probably the set up hassle of them and also the priming/preheating process is not great because we often use our stoves in our tent vestibules (naughty hunters).
Alright I've touched on the fundamental types/principles of stoves but be aware there is so many stoves out there now that combine aspects of each of these types I can't list them all. It's up to you to properly research any stove you may own or be thinking of purchasing to determine how it works exactly.
I'll do a big loop back to the start next and talk about micro-regulated stoves vs jets.Last edited by K95; 12-04-2020 at 02:26 PM.
-
12-04-2020, 02:12 PM #11
Damn. I didn't put in there roughly what each type of stove is good down to temperature wise.
I'll put it here before we go on.
These are rough numbers you can use if you can't be bothered doing the math yourself. Don't come to my house angry if your stove doesn't work when I said it should. Elevation effects temperature so be aware. Use good quality gas. These numbers are assuming you use some of the tricks I've said.
Upright canister stove : -7C
Inverted canister stove: -18C
Multi-fuel stove: colder than you can survive.
-
12-04-2020, 02:13 PM #12Banned

- Join Date
- Mar 2015
- Location
- North Canterbury
- Posts
- 5,462
So there is 'evaporative cooling' taking place inside the cylinder to use the correct term. Thankyou for posting as I would never have thought of that happening or your simple solution of a container of water, nor would I have thought about the ratio of gasses changing as gas is taken out. This is a really great learning session ! PS just read 3 and the term 'evaporative cooling' is there - I should have read before typing
-
12-04-2020, 03:25 PM #13
Part 5 - Regulated and Non regulated stoves.
This applies to upright canister stoves from part 2.
The two stoves shown both use a different type of valve to control the flow of fuel to the burner. The remote canister stove shown has what is called a 'Micro-regulator' while the other uses a basic needle type valve.
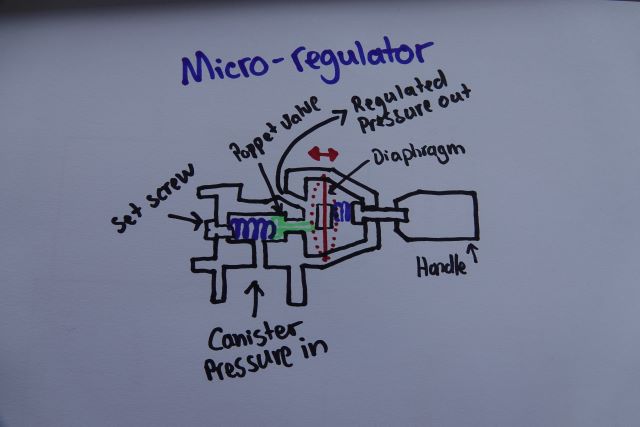
A micro-regulator stove will regulate the pressure supplied to the burner automatically so as the canister cools (evaporative cooling remember) the output will remain constant. That's quite handy. It means if we're using the stove at half throttle as it cools we don't have to manually open our valve more and more to keep the output the same. Each different brand will use a construction of their micro-regulator but I've drawn/copied a rough example above.
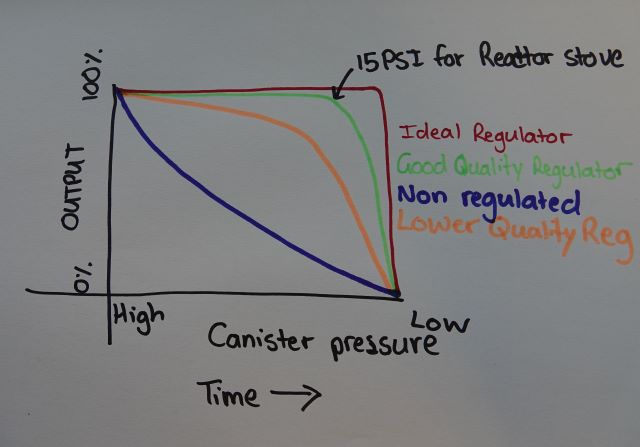
I have here an MSR Reactor stove, it's a micro-regulated type. MSR says it runs on max output at 15PSI. So we need at least 15PSI inside our canister to get full output from this stove. That's the limitation of a micro-regulator. Once our canister pressure falls below this pressure our micro-regulator stove is no better really than non-regulated upright canister stoves.
I've included another rough sketch based on MSR data to illustrate this.
Micro-regulators are great but just be aware they're not the be-all end-all of solving upright canister stove issues.

-
12-04-2020, 04:12 PM #14
-
12-04-2020, 04:36 PM #15
Part 6 - Safety.
I know, I know. Remember though, this is an open forum that anyone with access to the internet can see. I spent a lot of time when I was young eagerly browsing the forums sponging information. I need to put this in here. What may be blindingly obvious to you may not be to others with less experience.
I'm not going to list every DO NOT, there's enough of that in the manual for your stove. I'll just say a few things which sometimes aren't in there (directly mentioned anyway).
Connecting/disconnecting gas canisters.
Be careful/thoughtful when you are doing this in a hut. That Lindal valve I showed you in Part 1 has been known to get stuck in the open position before. Think candle lit hut and that canister pouring a massive amount of vapour out. Happens quicker than you can react. Be wise where you are connecting/disconnecting your stove.
Windshields.
Placing a metallic windshield around a cansiter can cause the canister to overheat and explode. The blue flame is in the vicinity of 1900C.
Oversize pots/pans
For similar reasons to windshields oversized pots and pans can reflect heat down onto a canister causing it to overheat and explode. This is a common cause of explosions with those benchtop type camping stoves that take the hairspray looking canisters.
Read your manual. Be sensible about how you use your gas stove and if you put some of this information into practice you'll get the most out of your stove not be left wondering what the hell is going on when things aren't working.
Similar Threads
-
Vargo Ti Triad/Triad Multi-Fuel Stoves
By Nesika in forum Gear and EquipmentReplies: 8Last Post: 10-03-2016, 10:56 PM
Tags for this Thread
Welcome to NZ Hunting and Shooting Forums! We see you're new here, or arn't logged in. Create an account, and Login for full access including our FREE BUY and SELL section Register NOW!!




 67Likes
67Likes LinkBack URL
LinkBack URL About LinkBacks
About LinkBacks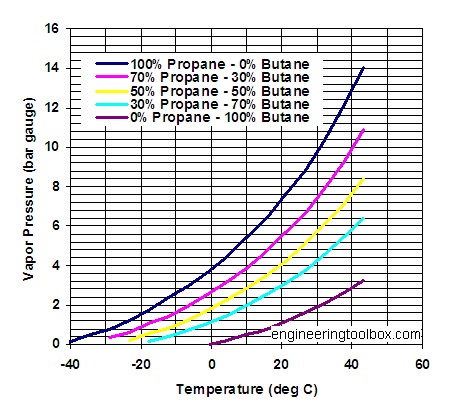




 Reply With Quote
Reply With Quote



Bookmarks